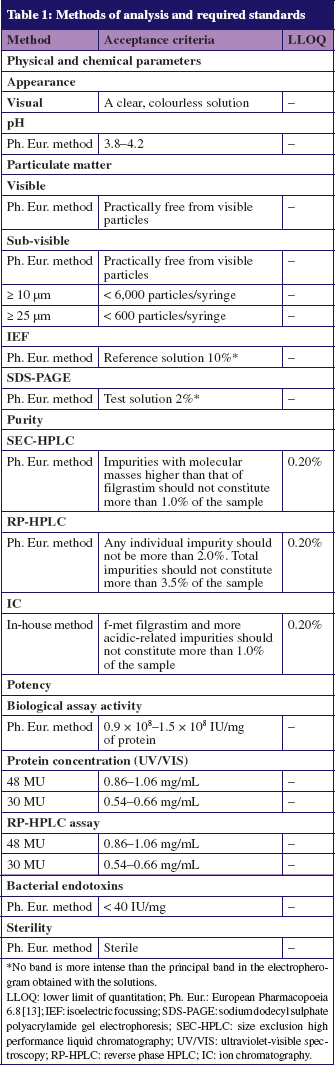
Filgrastim-Teva® seringues prêtes à l'emploi 30 MIU/0,5 ml et 48 MIU/0,8 ml sont remplacées à la PHEL par Zarzio® seringues

Real-world safety experience of tevagrastim/ratiograstim/biograstim and tbo- filgrastim, short-acting recombinant human granulocyte colony-stimulating factors | Request PDF

These highlights do not include all the information needed to use GRANIX safely and effectively. See full prescribing information for GRANIX.GRANIX® (tbo-filgrastim) injection, for subcutaneous use Initial U.S. Approval: 2012